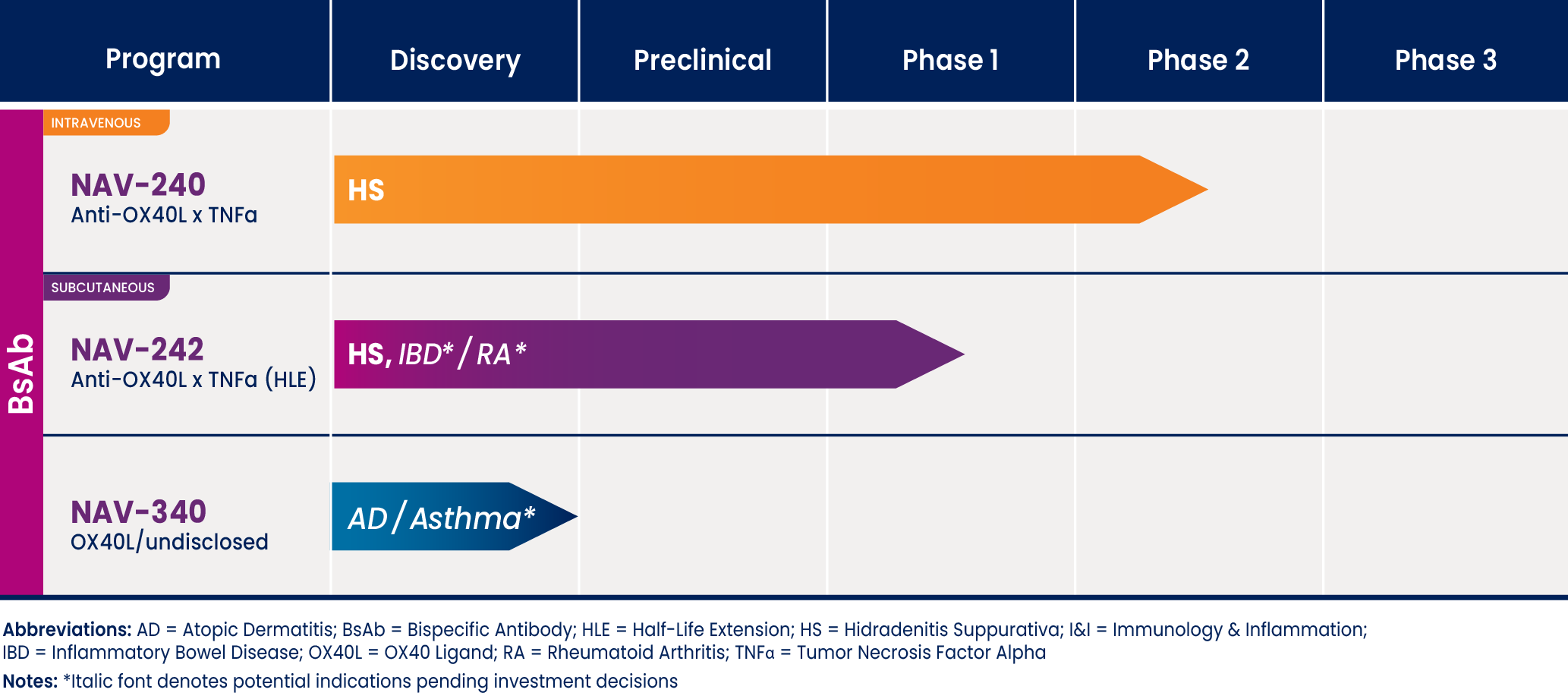
PIPELINE
Next-generation bispecific antibodies to raise the efficacy bar
At Navigator, we’re advancing a pipeline of expertly engineered bispecific antibodies that combine OX40L inhibition with other potentially synergistic mechanisms to deliver effective, well tolerated and convenient options for people living with complex inflammatory diseases
Hidradenitis suppurativa needs new treatment
Hidradenitis suppurativa or HS, is the first inflammatory disorder we are working to alleviate for patients through our pipeline.
HS is a chronic inflammatory skin condition that causes painful lumps, abscesses, and scarring to develop in areas where skin often rubs together like armpits, groin and under the breasts. When hair follicles become blocked, inflammation can start and deep, painful nodules can develop that eventually rupture and drain fluid. People with HS often experience symptom flare-ups that can significantly impact daily activities and their quality of life.
Over one million people in the U.S. live with moderate-to-severe HS which affects three times more women than men. Today, in the absence of effective treatment many patients experience delayed diagnosis and only 350,000 currently seek treatment, with many people struggling to manage their symptoms for years. The visible nature of the condition and its impact on intimate areas can profoundly affect self-esteem, relationships, and overall mental health, underscoring the critical need for effective new options.